The search for a cure to Alzheimer’s disease has long centred on a single culprit — amyloid plaques, sticky protein deposits that accumulate in the brain. Decades of plaque-focused trials have produced modest clinical gains at enormous cost, with the development of a single new Alzheimer’s drug costing, on average, over $1.2bn. Between 1996 and 2021, $42.5bn of private capital was spent on Alzheimer’s research and drug discovery, yielding only five novel drugs.
Researchers at Rice University in Houston have now upended that assumption, using label-free laser-imaging and artificial intelligence to reveal, in a mouse model, that Alzheimer’s is a disease of widespread chemical disruption, not a single protein defect.
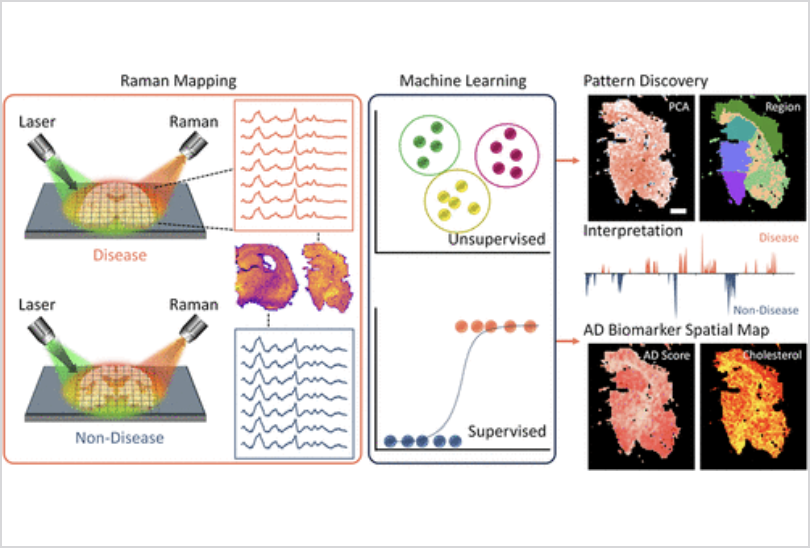
Employing hyperspectral Raman imaging, a technique that detects the unique chemical signatures of molecules without dyes or stains, the Rice researchers scanned entire brains slice by slice to construct high-resolution molecular maps of both healthy and diseased tissue in its natural state.
The maps revealed patterns invisible to conventional imaging. “We found that the changes caused by Alzheimer’s disease are not spread evenly across the brain,” said Ziyang Wang, a co-author of the study, clarifying why some symptoms develop slowly and single-target treatments yield only modest results. The researchers found that chemical changes linked to Alzheimer’s extend to levels of cholesterol, essential for maintaining healthy brain cell structure, and glycogen, a local energy reserve. The largest difference was found in the hippocampus and cortex, areas linked to memory and cognition.

“Together, these findings support the idea that Alzheimer’s involves broader disruptions in brain structure and energy balance, not only protein buildup and misfolding,” said Shengxi Huang, an associate professor of electrical & computer engineering and a corresponding author on the study.
A fuller molecular map of the Alzheimer’s brain could redirect capital toward multi-target therapies, diversifying drug development pipelines that have long been concentrated on proteins.
The volume of data produced required a new approach to analysis; machine learning (ML) models were used to create the chemical map of the Alzheimer’s brain. Unsupervised ML models categorised the tissue based on molecular characteristics, whilst supervised ML models, trained on known samples, revealed which areas of the brain had chemical changes most characteristic of the disease. By narrowing the field of potential targets before expensive human trials begin, ML may reduce late-stage failure, the costliest bottleneck in pharmaceutical development, and pave the way for more affordable, effective treatments.
The urgency is not merely medical. Alzheimer’s and other forms of dementia already cause more deaths globally than breast and prostate cancer combined and, in Britain alone, cost the economy £42bn a year, according to Alzheimer’s Research UK. With related deaths projected to quadruple by 2040, the question of whether science can find treatments that work, and that health systems can afford, is becoming one of the defining pressures of ageing societies.
The Rice atlas does not deliver a cure. But for a disease that has resisted medicine’s best efforts for decades, knowing where to look may matter as much as knowing what to do — and science finally has somewhere new to point.
View the full research paper through this link.